About SORAPEX
Profile
In 2012 German pharmacist Beate Sonntag moved from Hannover, Germany to Richmond upon Thames, United Kingdom. After arriving and settling the wish to engage in meaningul work and the new inspiring environment of Greater London led to the decision to create and establish SORAPEX Ltd. in England. Soon, SORAPEX Ltd. was successfully providing professional services for global regulatory affairs to the pharmaceutical industry.
The relocation back to Hannover, Germany in 2016 (early BREXIT, kind of) led to the business decision to dissolve the company in the UK and to continue the business operating as a professional freelancer (Freiberufler) in Germany. However, except for the location and the legal framework, neither the business model or services of the company nor its client base did change. The name and domain "SORAPEX" remained in use, the description “Regulatory Affairs Services for Pharmaceuticals” was complemented.
Because of its success and growing client base, in 2019 the legal framework for the business was changed again and the company is now incorporated and officially registered in Germany as SORAPEX GmbH, with Beate Sonntag as its founder, owner and CEO.
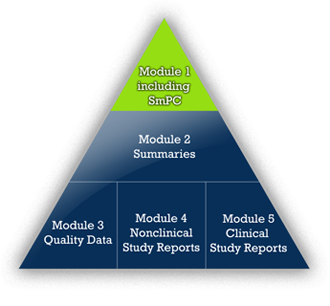
Name
The idea for the company's name derives from APEX. Originally Latin, the term APEX is known in German, English, and French. It describes the localization of the labeling information in the top triangle of the ICH Common Technical Document (CTD), Module 1.3.
The SmPC is the "ultimate" summary of the CTD.
Presenting the individual motivation of the founder and her focus on regulatory affairs, SORAPEX communicates the message of SOnntag - Regulator Affairs Pharmaceutical EXcellence.
Slogan
SORAPEX' slogan "Always lead the label" is aiming to summarize the high importance of labeling, especially within the Summary of Product Characteristics (SmPC) in the context of the well-known phrase "always read the label" as used during the promotion of a product:
- The SmPC is the basis of information for health care professionals on how to use the medicinal product safely and effectively
- The SmPC is the key labeling document for the establishment of the patient information leaflet informing patients about the correct use of the medicinal product.
- The SmPC is one of the main documents in the public domain.
- The SmPC is published in national compendia in the corresponding national language (e.g. Vidal, Arzneimittel-Kompendium and eMC).
- The SmPC is the agreed position between the marketing authorization holder and the regulatory authority.
- The SmPC is the basis for any promotional message and is key to reimbursement.
- Proper labeling of products regulated by the FDA is extremely crucial. Improper labeling could cost a company millions of dollars in litigation and bad publicity.







